
Introduction
Liquid dropout in a pipeline doesn't announce itself — it just starts causing problems. Hydrocarbon dew point (HCDP) calculation tells you exactly where that threshold is: the temperature at which the heaviest components in a gas mixture begin to condense from vapor at a given pressure.
Pipeline operators, process engineers, lab technicians, and emissions monitoring professionals rely on accurate HCDP calculations to prevent liquid formation, protect equipment, and satisfy gas quality specifications in custody transfer agreements.
Once temperatures drop below the HCDP, hydrocarbon liquids accumulate in pipelines — increasing pressure drop, reducing line capacity, and threatening compressors and turbines. Research from Michell Instruments shows that natural gas-fueled turbines need at least 50°F of superheat above the HCDP to prevent flame flashback during expansion. Miss that margin, and the repair costs are severe.
Getting the HCDP right depends on more than a formula — it requires accurate gas composition data, the right equation of state, and an understanding of where the calculation can mislead you.
This guide covers all of it: what HCDP calculation involves, how C6+ characterization affects results, which equations of state are used, and when to calculate versus measure directly.
TL;DR
- HCDP calculation determines condensation temperature using gas chromatograph composition data combined with an equation of state model
- Peng-Robinson (PR) and Soave-Redlich-Kwong (SRK) are the two most accepted equations; SRK typically yields slightly higher predicted dew points
- Accuracy depends heavily on C6+ characterization; lumped fractions with fixed split ratios introduce errors exceeding 70°F
- Calculation is preferred when HCDP values at multiple pressures or the cricondentherm are needed from a single analysis
- A validated, properly calibrated GC paired with NIST-traceable reference standards is the starting point for reliable HCDP results — SpecGas Inc. supplies precision calibration gas mixtures built to that standard
What Is Hydrocarbon Dew Point Calculation?
HCDP calculation uses the known mole fractions of each component in a gas mixture—fed into a thermodynamic equation of state—to predict the pressure-temperature combination at which the first droplet of liquid hydrocarbon condenses from the vapor phase.
The calculation produces a phase envelope showing the boundary between all-vapor and two-phase conditions, as well as the cricondentherm—the maximum temperature at which condensation can occur regardless of pressure. Together, these outputs let operators assess gas behavior across the full operating range without running separate tests at each pressure condition.
That stands in contrast to chilled mirror analyzers, which physically cool the gas until condensation is visually or optically detected. Rather than observing condensation in real time, the calculation derives the dew point computationally from composition—making it practical for evaluating multiple pressure scenarios from a single analysis.
Why HCDP Calculation Matters for Gas Mixtures
Operational Consequences of Liquid Dropout
Hydrocarbon liquids forming in gas systems create serious operational problems:
- Increased pressure drop and reduced line capacity
- Damage to compressors and gas turbines
- Pressure regulator freezing
- Measurement errors in custody transfer flow meters
- Fouled metering stations requiring frequent pigging operations
These risks are precisely why pipeline operators and regulators have moved to enforce strict HCDP limits as a condition of delivery.
Contractual and Regulatory Drivers
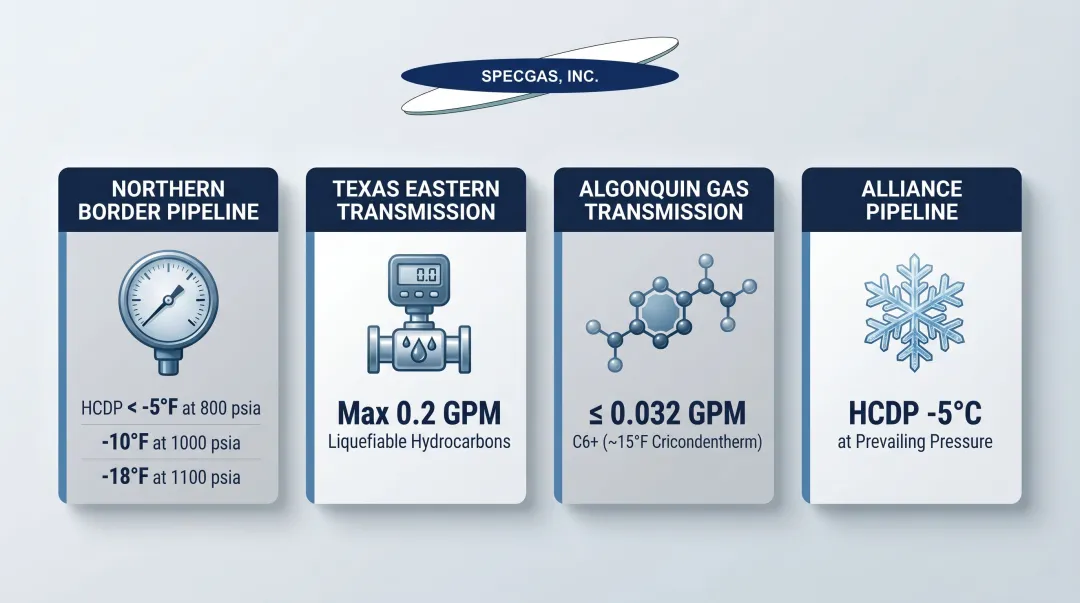
Pipeline tariff specifications increasingly enforce strict HCDP limits as conditions of delivery. The Federal Energy Regulatory Commission (FERC) recognizes HCDP and C6+ GPM limits as valid methods to control hydrocarbon liquid dropout in jurisdictional tariffs. Common specifications include:
- Northern Border Pipeline: HCDP less than -5°F at 800 psia, -10°F at 1000 psia, or -18°F at 1100 psia
- Texas Eastern Transmission: Maximum 0.2 GPM of liquefiable hydrocarbons
- Algonquin Gas Transmission: ≤0.032 GPM C6+ (correlates to ~15°F cricondentherm)
- Alliance Pipeline (Canada): HCDP specification of -5°C at prevailing operating pressure
Key Advantage Over Physical Measurement
A single GC analysis combined with an equation of state can produce three critical outputs from one measurement:
- HCDP at any pipeline pressure
- The full phase envelope
- The cricondentherm
That means operators can verify gas quality across different line pressures from a single analysis — no need for multiple physical analyzers spread across the line.
How the HCDP Calculation Process Works
The process starts with detailed gas composition from a GC analysis, uses a thermodynamic model to predict vapor-liquid equilibrium behavior, and iterates to find the exact temperature at which the vapor fraction equals 1.0 at the specified pressure.
Both Peng-Robinson and SRK equations are cubic equations of state that account for real gas behavior using each component's critical temperature, critical pressure, and acentric factor, plus binary interaction parameters (BIPs) for component pairs. The accuracy of these parameters directly influences the reliability of dew point predictions.
Critical validation requirement: Operators must validate EOS software against measured experimental dew point data before use. Different software implementations of the same EOS can produce differing results depending on BIP sets and component property databases used.
Step 1: Gas Composition Analysis
Obtain a detailed GC analysis of the gas mixture, ideally analyzing individual components through at least C9 and preferably C12. The GC must be properly calibrated using representative reference calibration gases.
NIST-traceable specialty gas standards—such as those from SpecGas Inc.—minimize compositional uncertainty in the heavy hydrocarbon fractions that most strongly influence HCDP. SpecGas produces stable low PPM and PPB concentration hydrocarbon calibration mixtures using proprietary gravimetric blending techniques and cylinder treatment processes designed to enhance stability.
Step 2: C6+ Fraction Characterization
Since field GCs often report all hexane-and-heavier components as a single lumped "C6+" value, a characterization method must distribute this fraction into individual component amounts.
Critical finding: Common fixed-ratio splits such as GPA's 60/30/10 C6/C7/C8 split introduce large errors and should be avoided for HCDP work. SwRI research confirms that using a lumped C6+ characterization instead of full characterization can change the computed dew point by as much as 70°F, consistently producing under-predictions of that magnitude.
Extended analyses through C9+ or Gaussian-based characterization methods improve accuracy substantially. ISO 23874 specifies that GC systems for HCDP calculation must measure all hydrocarbons from C5 to C12 — making full characterization the required baseline, not an optional refinement. That complete compositional picture then feeds directly into the EOS calculation.

Step 3: EOS Calculation and Dew Point Output
Feed the characterized composition into the EOS software at the target pressure(s). The software iterates to find the dew point temperature and can generate the full phase envelope including the cricondentherm.
EOS selection affects results in a predictable way:
- SRK predicts slightly higher dew points than PR for the same composition — a conservative result that suits pipeline safety applications
- Peng-Robinson tends to predict marginally lower dew points, which can under-protect in critical operating scenarios
For most custody transfer and pipeline integrity applications, the conservative SRK output is the preferred choice.
Key Factors That Affect HCDP Calculation Accuracy
Four variables drive most of the uncertainty in HCDP calculations. Understanding each one helps engineers select the right analytical methods and avoid systematic errors.
C6+ characterization method: This is the single largest source of error in HCDP calculations. Using a fixed C6/C7/C8 ratio instead of an extended C9+ analysis can produce errors exceeding 70°F. SwRI research shows that obtaining composition data through C9 allows prediction of true hydrocarbon dew points to within ±25°F in many cases.
GC instrument accuracy and calibration: Field GCs have limited precision for heavy hydrocarbon components, typically at best ~2% of range error for C6+. Just 1 ppmV of a a C10 component can shift the calculated HCDP by as much as 10°F. Accurate calibration with high-purity, NIST-traceable reference gas standards is essential to minimize this uncertainty. SpecGas Inc. produces NIST-traceable calibration standards specifically suited to this application.
Choice of EOS and binary interaction parameters: Many BIP sets are not validated across the full range of pipeline pressures and temperatures, especially for heavier C6+–C6+ pairs. A documented lack of experimental data for heavy-heavy interactions causes many software packages to default these BIPs to zero, which can cause systematic underprediction of HCDP in richer gases.
Sampling integrity: Collecting and conditioning a gas sample for GC analysis can alter its composition, particularly if heavy components drop out during pressure reduction. Pressure reduction causes rapid cooling due to the Joule-Thomson effect — roughly 7°F per 100 psi reduction. Improper sampling is a primary cause of underestimated HCDP values in field applications.
Common Issues and Misconceptions in HCDP Calculation
Conflating Dew Point with Condensation Rate
The calculated HCDP marks the thermodynamic onset of condensation (zero liquid) but gives no information about how much liquid actually forms with further cooling. A gas can technically be below its HCDP but still carry negligible liquid.
Industry research established the concept of a "practical HDP" based on a tolerable liquid threshold. Studies on the Questar Pipeline demonstrated that a liquid dropout level of 0.002 gallons per thousand cubic feet (GPM) has a negligible effect on pressure drop and does not disrupt pipeline operations.
Assuming Any C6+ GC Analysis Is Sufficient
Using a standard C6+ GC with assumed fixed split ratios for HCDP calculations is a common source of significant errors. For acceptable accuracy, extended analysis is required — at minimum through C9, and ideally through C12. Key requirements include:

- Extended GC analysis to at least C9 (C12 preferred for higher accuracy)
- Verified split ratios rather than assumed fixed values
- Recalibration when feed composition shifts significantly
Relying on a Single Calculated HCDP Value
The dew point is pressure-dependent, and the phase envelope has a complex shape — including a retrograde condensation region where liquid can form as pressure increases. A single HCDP value captures only one point on that curve. Operators should calculate and review the full dew point curve across their operating pressure range, not just at one condition.
When Gas Composition Is Poorly Characterized
When a gas stream has highly variable heavy hydrocarbon content, a single EOS calculation based on one GC snapshot may not represent actual operating conditions. Changing production conditions can shift the heavy-end composition enough to invalidate earlier results. Periodic recalibration and extended analyses are required to keep calculations reliable.
Frequently Asked Questions
How to calculate the dew point of a gas?
HCDP is calculated by inputting the gas mixture's component mole fractions from a GC analysis into an equation of state such as Peng-Robinson or SRK, then solving iteratively for the temperature at which the vapor fraction equals 1.0 at a specified pressure. An extended C9+ composition analysis measurably improves accuracy.
What is the difference between hydrocarbon dew point and cricondentherm?
The HCDP is the condensation temperature at a specific pressure, while the cricondentherm is the maximum temperature at which condensation can occur at any pressure. Above the cricondentherm, the gas mixture remains entirely in the vapor phase regardless of pressure.
Which equation of state is most accurate for HCDP calculation?
Both Peng-Robinson (PR) and Soave-Redlich-Kwong (SRK) are widely used — SRK tends to predict slightly higher dew points, making it a common choice for conservative pipeline safety work. In practice, accuracy depends more on C6+ characterization and validated binary interaction parameters than on which EOS is selected.
Why does C6+ fraction characterization matter so much for HCDP calculation?
Heavier hydrocarbons (C6+) have a disproportionately large influence on the dew point despite being present in small concentrations. Errors in how the C6+ fraction is split into individual components can produce HCDP errors of 30–70°F or more, making an extended GC analysis through C9+ or higher critical for reliable results.
When should direct measurement be used instead of EOS calculation for HCDP?
Chilled mirror measurement is the better choice when gas composition is unknown, highly variable, or difficult to sample for lab analysis, and when on-stream validation is required. EOS calculation is preferable when HCDP values across multiple pressures or the cricondentherm must be derived from a single analysis.
What operational problems does liquid hydrocarbon dropout cause in gas systems?
Hydrocarbon liquid in pipelines increases pressure drop, reduces line capacity, damages compressors and turbines, causes pressure regulator freezing, and creates significant errors in custody transfer flow measurement—making HCDP control a critical operational and commercial issue.


