
Introduction
Dimethyl sulfide (DMS) is a reactive sulfur compound that requires precise calibration gas standards for accurate measurement in environmental monitoring, emissions testing, atmospheric research, and industrial process control. Calibration errors directly affect compliance reporting and scientific data quality. A 1986 peer-reviewed study found that incorrect DMS calibration using GC/FPD produced results deviating from true values by 36–206%.
This guide covers what DMS is, why calibration accuracy matters, how standards are prepared, the cylinder stability challenges DMS presents, and which analytical methods and regulations govern its measurement.
Key Takeaways
- Calibration errors for DMS can skew analytical results by up to 206% — accurate standards are non-negotiable
- Cylinder passivation is essential: treated cylinders maintain DMS stability for over 60 months; untreated cylinders can fail within weeks
- Regulatory Method 16 requires strict TRS calibration performance: ≤5% drift, 50 ppb sensitivity
- Gravimetric preparation with NIST traceability provides the foundation for primary standard gas mixtures
- Target concentration range matters: atmospheric research requires sub-nmol/mol standards, while industrial emissions monitoring operates at ppm levels
What Is Dimethyl Sulfide and Why Is It Measured?
Chemical Identity and Physical Properties
Dimethyl sulfide (chemical formula (CH₃)₂S, CAS No. 75-18-3) is a volatile organosulfur compound with a molecular weight of 62.134 g/mol and a boiling point of 37.3°C. It has a distinctive, pungent odor often described as cabbage-like or sulfurous, with humans detecting it at concentrations as low as 1–20 ppb.
DMS must be distinguished from related compounds:
- Dimethyl sulfoxide (DMSO): The oxidized form of DMS, formula (CH₃)₂SO, commonly used as a pharmaceutical solvent
- Dimethyl disulfide (DMDS): A different organosulfur compound with two sulfur atoms
Sources and Environmental Significance
DMS originates from both natural and industrial sources:
Natural Sources:
- Marine environments produce DMS from dimethylsulfoniopropionate (DMSP) in phytoplankton, making it the main natural source of atmospheric sulfur
- Soils and certain foods/beverages (including beer, where it registers as an off-flavor compound)
- Ambient atmospheric mixing ratios range from 46 ppt to 125 ppt in remote marine boundary layers
Industrial Sources:
- Kraft pulp mills
- Petroleum refineries
- Wastewater treatment facilities
- Food and beverage production
Regulatory and Scientific Drivers
Knowing where DMS comes from directly shapes why it needs accurate measurement. DMS is one of four Total Reduced Sulfur (TRS) compounds regulated under Method 16 for stationary source emissions:
- H₂S (hydrogen sulfide)
- Methyl mercaptan
- DMS (dimethyl sulfide)
- DMDS (dimethyl disulfide)
Atmospheric chemistry networks monitor DMS globally because its oxidation yields sulfur dioxide, methanesulfonic acid, and sulfuric acid. These compounds form sulfate aerosols that act as cloud condensation nuclei and influence Earth's radiative balance.
Why Accurate DMS Calibration Gas Standards Are Critical
The Calibration Principle
A calibration gas standard provides a known concentration of DMS against which an analytical instrument's response is normalized. Without an accurate standard, every downstream measurement carries unquantifiable error.
Real-World Calibration Failures
Research in beer quality analysis demonstrated the dramatic impact of calibration methodology. Using an incorrect calibration approach for DMS with GC/FPD led to results differing from true values by 36–206%. This error stemmed from differential quenching responses caused by co-eluting non-sulfur compounds—a clear case for matrix-matched or validated calibration techniques.
NIST Traceability Requirements
Metrological traceability means a measurement result relates to a reference through an unbroken chain of calibrations, each contributing to measurement uncertainty. For DMS standards:
- Primary Standard Gas Mixtures (PSMs): Prepared gravimetrically and traceable to SI units per ISO 6142-1:2015
- Working Standards: Commercial mixtures certified against PSMs for routine instrument calibration
- Expanded Uncertainty: Reported with coverage factor k=2 at ~95% confidence interval
The accuracy of PSMs determines the ceiling of accuracy for all downstream measurements. SpecGas Inc. blends all DMS calibration mixtures gravimetrically in-house, with NIST traceability verified at each step—an approach rooted in Alfred Boehm's R&D work with Messer Griesheims Industries dating back to 1976.
Application-Specific Concentration Ranges
That accuracy ceiling only holds if the standard matches the actual measurement range. DMS monitoring spans widely varying concentration levels:
- Sub-nmol/mol (ppt): Atmospheric and oceanic research
- 50 ppb to 100 ppmv: Industrial TRS emissions testing under Method 16
- ppb levels: Ambient air quality monitoring
Calibration standards must match the expected concentration range of the application. Using a 100 ppm standard to calibrate an instrument measuring 50 ppt ambient levels introduces unacceptable uncertainty.
How DMS Calibration Gas Standards Are Prepared
Gravimetric Preparation (Primary Method)
ISO 6142-1:2015 specifies gravimetric preparation for Class I calibration gas mixtures. DMS is weighed and introduced into a pressurized cylinder with a balance gas (typically nitrogen or clean air) using precise mass measurements. Concentration is calculated from mass ratios, providing direct traceability to SI units.
Key requirements:
- High-accuracy molar mass determination
- Purity analysis per ISO 19229
- Accounting for impurities in parent gases
- Verification that added masses convert accurately to amounts of substance
Method 16 Calibration Sources
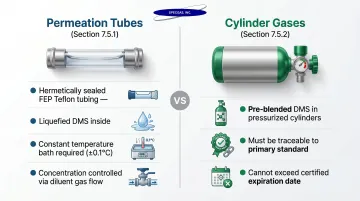
Method 16 Section 7.5 specifies two acceptable calibration approaches:
1. Permeation Tubes (Section 7.5.1):
- Hermetically sealed FEP Teflon tubing containing liquefied DMS
- Constant, temperature-dependent permeation rate
- Requires constant temperature bath (±0.1°C)
- Known concentration generated by controlling diluent gas flow rate
2. Cylinder Gases (Section 7.5.2):
- Pre-blended DMS mixtures in pressurized cylinders
- Must be traceable to a primary standard
- Cannot be used beyond certified expiration date
Analytical Verification
Before certification, prepared standards must be verified by independent analytical methods (typically GC with FPD or mass spectrometry), confirming the gravimetrically calculated concentration matches the analytical measurement within acceptable expanded uncertainty. This step prevents release of standards with preparation errors or undetected contamination.
Internal Standards for Low-Concentration Calibration
Analytical verification addresses batch-level accuracy. At trace levels, internal standards go further by compensating for ongoing matrix effects and instrument drift:
- Seawater analysis: Deuterated DMS-d6 achieves precision <2% at 0.1 nM concentrations
- Atmospheric analysis: Isotope-labeled CD₃SCD₃ in APCI-MS enables continuous underway shipboard measurements
Tracking the analyte-to-internal-standard ratio over time reveals instrument drift before it affects reported concentrations — a practical safeguard for long-duration monitoring campaigns.
The Stability Challenge: DMS Adsorption and Cylinder Selection
The Adsorption Problem
DMS is chemically reactive and prone to adsorption loss on metal and untreated cylinder surfaces. Even a gravimetrically accurate mixture can degrade after filling if the cylinder's internal surface isn't properly passivated—meaning instruments get calibrated against concentrations lower than labeled.
Comparative Stability Data
A 2018 peer-reviewed study in Metrologia evaluated adsorption loss of 6 nmol/mol DMS mixtures in three cylinder types:
| Cylinder Treatment | Result |
|---|---|
| Untreated Aluminum | Substantial adsorption loss (peak area ratios <0.72) |
| Aculife IV+III | Substantial adsorption loss (peak area ratios <0.96) |
| Experis-treated | Negligible adsorption loss; stable >60 months within 3% expanded uncertainty |

Cylinder Passivation Technology
Proprietary internal surface treatments create a chemically inert barrier between reactive DMS gas and the cylinder wall. This process directly determines both fill accuracy and long-term shelf life — and it requires specialized expertise that most general gas suppliers don't carry.
SpecGas Inc. employs a proprietary internal cylinder treatment process developed specifically for reactive gas mixtures like DMS, backed by the SpecGas Stability Guarantee. Founder Alfred Boehm built this expertise at Messer Griesheim Industries in Germany, then continued that R&D work after transferring to the US in 1991 — focusing specifically on internal cylinder treatment for reactive gases before founding SpecGas in 2001.
Analytical Detection Methods and Regulatory Requirements for DMS
Gas Chromatography with Flame Photometric Detection (GC/FPD)
GC/FPD is the primary technique for DMS measurement. The FPD's selective response to sulfur-containing compounds makes it ideal, but requires systematic interference management.
Method 16 interference controls:
- Moisture (Section 4.1): Prevented by maintaining probe and filter box ≥120°C
- CO and CO₂ (Section 4.2): Chromatographically eluted before sulfur compounds to prevent FPD quenching (desensitization)
- Particulate Matter (Section 4.3): Eliminated using Teflon filter
- SO₂ (Section 4.4): Removed selectively using citrate buffer scrubber
Method 16 Performance Thresholds
Method 16 governs semicontinuous determination of TRS from stationary sources:
- Sensitivity: 50 ppb minimum using 10 mL sample loop
- Analytical Range: 0.1 to 100 ppmv for industrial sources
- Calibration Drift: ≤5% between pre- and post-run checks
- Analytical Precision: Triplicate injections must not vary >5% from mean

Research-Grade Trace Detection
Method 16 addresses industrial source emissions, but atmospheric and environmental DMS monitoring operates at sub-nmol/mol levels — well below the ~1 ppb floor of standard GC-FPD. Research applications require more sensitive techniques:
- Atmospheric Pressure Chemical Ionization Mass Spectrometry (APCI-MS): Achieves 220 ppt detection limits with 1-minute averaging
- Purge-and-Trap GC-MS: For seawater analysis
- Specialized calibration standards: Certified at ultra-low levels with appropriately low expanded uncertainties
Each of these methods demands calibration standards certified at the concentration level being measured — making traceability and low expanded uncertainty non-negotiable for credible results.
Frequently Asked Questions
Are DMS and DMSO the same?
No. DMS (dimethyl sulfide, (CH₃)₂S) and DMSO (dimethyl sulfoxide, (CH₃)₂SO) are chemically distinct. DMSO is the oxidized form of DMS and serves as a common laboratory solvent. DMS is a volatile gas with a strong odor, relevant to atmospheric chemistry and emissions monitoring.
Where is dimethyl sulfide found on Earth?
DMS occurs naturally in marine environments (produced by phytoplankton from DMSP), in soils, and in certain foods. It's also released as an industrial byproduct from kraft pulp mills, petroleum refineries, and wastewater treatment facilities—all sources subject to emissions monitoring and regulatory oversight.
What does DMS smell like?
DMS has a distinctive, pungent odor often described as cabbage-like, sulfurous, or reminiscent of the sea. Humans can detect it at concentrations as low as 1–20 ppb, with a mean odor detection threshold of 11 ppb. At those detection thresholds, even minor fugitive emissions from industrial sources can trigger odor complaints, which is why regulatory limits and continuous monitoring programs exist.
How do you test for dimethyl sulfide?
DMS is most commonly measured using Gas Chromatography with Flame Photometric Detection (GC/FPD), which selectively detects sulfur-containing compounds. At very low environmental concentrations, purge-and-trap GC-MS or APCI-MS methods are used. All methods require a certified DMS calibration gas standard to produce accurate quantitative results.
What concentration levels are DMS calibration gas standards available in?
DMS standards span a wide range depending on application: from sub-nmol/mol (parts-per-trillion) levels for atmospheric and oceanic research, to ppb levels for ambient air monitoring, up to ppm levels for industrial TRS emissions testing under Method 16. The standard must be matched to the expected concentration range in the sample matrix.
How long are DMS gas standards stable in a cylinder?
Stability depends on cylinder treatment. In properly passivated cylinders, DMS standards at low nmol/mol levels can remain stable for over 60 months within 3% uncertainty. Untreated cylinders can lose significant concentration within days due to surface adsorption. Always verify the certified expiration date and use within that window. SpecGas backs its DMS mixtures with a Stability Guarantee, backed by proprietary cylinder treatment technology.


