
Introduction
According to the 2023 Annual Report of the National Poison Data System, there were over 2 million human poison exposures in the United States, with household cleaning substances accounting for 7.12% of all cases. More concerning, a peer-reviewed analysis of poisoning data from 2015 to 2022 identified 75,186 chlorine and chloramine gas exposures, with 85.5% occurring in homes. These incidents resulted in 16.7% of victims requiring emergency department care, 2% needing hospital admission, and 26 deaths.
These numbers don't reflect careless behavior. Most victims simply didn't know that combining common household cleaners can produce toxic gases. Bleach (sodium hypochlorite) is safe when used alone and as directed.
The risk starts the moment it contacts certain other products. That contact triggers chemical reactions releasing gases capable of causing respiratory injury, hospitalization, and death.
This guide identifies the specific products you should never mix with bleach, explains what happens chemically when you do, and covers how to respond if exposure occurs.
Key Takeaways
- Never mix bleach with ammonia, acids (vinegar, toilet bowl cleaners), or rubbing alcohol—each combination produces a different toxic gas
- The main hazards: chloramine gas (bleach + ammonia) and chlorine gas (bleach + acids)—both cause serious respiratory injury
- Symptoms range from eye and throat irritation to chest pain, fluid in the lungs, and respiratory failure
- If exposed, leave the area immediately, get fresh air, and seek medical attention for anything beyond mild irritation
- Always read product labels before use and store incompatible chemicals separately
Dangerous Bleach Combinations and the Gases They Produce
Bleach's chemical name is sodium hypochlorite (NaOCl), and its oxidizing nature makes it highly reactive. When it contacts acids, nitrogen-containing compounds like ammonia, or certain solvents, it rapidly releases toxic byproduct gases. These reactions happen fast—often within seconds of contact—and the gases produced can cause immediate harm.
Bleach and Ammonia: Chloramine Gas
When sodium hypochlorite reacts with ammonia (NH₃), it forms chloramine gases: primarily monochloramine and dichloramine. These volatile compounds are toxic upon inhalation and cause serious respiratory irritation and lung injury.
Hidden sources of ammonia include:
- Glass and window cleaners (e.g., Windex Original contains ammonium hydroxide)
- Multi-surface sprays and all-purpose cleaners
- Interior and exterior paints (some brands contain 0.1–1% ammonium hydroxide)
- Urine (making toilet cleaning with bleach extremely dangerous)
Chloramine gas is less volatile than chlorine gas, but that doesn't make it safer. It causes shortness of breath, chest tightness, nausea, watery eyes, throat irritation, and wheezing, and prolonged exposure can lead to pneumonia and fluid in the lungs.
Peer-reviewed case reports document life-threatening toxic pneumonitis requiring prolonged hospitalization after individuals mixed household ammonia and bleach.
Bleach and Acids: Chlorine Gas
When bleach contacts any acidic cleaner, the pH drops below 4, triggering the release of chlorine gas (Cl₂). This gas then reacts with moisture in the air and lungs to form hydrochloric and hypochlorous acids, which cause tissue damage on contact.
Everyday acid-containing products people routinely use alongside bleach:
- Vinegar (acetic acid)
- Toilet bowl cleaners (hydrochloric acid)
- Drain cleaners (sulfuric acid)
- Rust removers (phosphoric acid)
- Automatic dishwasher detergents
- Brick and concrete cleaners
- Some glass cleaners
Even small amounts of residual acid from a previously used product can trigger a reaction. A 1991 CDC report documented five episodes of chlorine gas toxicity at state hospitals caused by mixing bleach with a 4% phosphoric acid cleaner, causing eye irritation, nausea, dizziness, and chest tightness in at least 14 people.
Accidental sequential use is just as dangerous as deliberate mixing: cleaning a surface with an acid-based product and then applying bleach without rinsing can produce the same toxic result.
Bleach and Other Products
Beyond ammonia and acids, several other common products react dangerously with bleach:
- Rubbing alcohol: Produces chloroform and other chlorinated compounds, both highly toxic
- Hydrogen peroxide: Triggers an oxygen-releasing reaction that accelerates oxidation and can rupture containers or cause splashing
- Oven cleaners: React with bleach to release harmful fumes; never use them in sequence
- Certain insecticides: Can form toxic byproducts when combined with sodium hypochlorite
- Pool chemicals (calcium or sodium hypochlorite): Must never be mixed with each other or with any household cleaner

Health Effects of Toxic Gas Exposure
Chlorine and chloramine gases target the respiratory airways and mucous membranes. Chlorine gas has a relative density of approximately 2.47 to 2.48, making it heavier than air — it settles in low-lying, poorly ventilated spaces like bathrooms and basements. That's why getting out immediately is critical: the gas pools where you're standing.
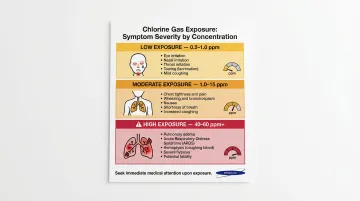
Chlorine Gas Exposure Symptoms by Concentration
Low concentrations (0.2–1.0 ppm):
- Eye, nose, and throat irritation
- Tearing and watery eyes
- Coughing and runny nose
- Detectable pungent, irritating odor
Moderate levels (1.0–15 ppm):
- Chest pain and shortness of breath
- Wheezing and difficulty breathing
- Vomiting and nausea
- Persistent coughing
High concentrations (40–60 ppm and above):
- Pulmonary edema (fluid in the lungs)
- Acute respiratory distress syndrome (ARDS)
- Hemoptysis (coughing up blood)
- Potential death
The NIOSH Immediately Dangerous to Life or Health (IDLH) limit is 10 ppm. At concentrations as low as 1 ppm, perceivable sensory irritation begins. Critically, symptoms can occur even at concentrations below the detectable odor threshold of approximately 3.5 ppm.
Chloramine Gas Symptoms
Chlorine isn't the only concern — chloramine gas poses its own serious risks. Exposure causes shortness of breath, chest tightness, nausea, watery eyes, throat irritation, and wheezing. Prolonged exposure can lead to pneumonia and fluid in the lungs. Though chloramine is less volatile than chlorine gas, it remains a dangerous inhalation hazard at household exposure levels.
Populations at Heightened Risk
People with asthma, COPD, or other pre-existing respiratory conditions are far more vulnerable to even brief or low-level exposures. Children and elderly individuals also face significantly higher risk due to smaller lung capacity and potentially compromised immune systems.
No Antidote Exists
There is no specific antidote for chlorine or chloramine gas inhalation. Treatment is supportive, relying on fresh air, oxygen, bronchodilators for wheezing, and in severe cases, mechanical ventilation. Once exposure occurs, management is reactive — accurate gas detection beforehand is what keeps workers safe.
Facilities that monitor for chlorine and other toxic gases depend on detector accuracy, which starts with properly calibrated equipment. SpecGas Inc. provides NIST-traceable chlorine calibration gas standards in concentrations from 0.5 ppm to 10%, giving industrial sites and research labs a verified reference point for gas detector calibration across the full range of exposure risk.
Safe Handling, Storage, and Workspace Practices
Following baseline safety rules prevents accidental chemical mixing and protects you from toxic gas exposure.
Safe bleach use rules:
- Read every product label before use
- Never combine products in the same bucket, mop, or spray bottle
- Use one product at a time and rinse surfaces thoroughly before applying another
- Always work in a well-ventilated area with windows open or exhaust fans running
- Wear rubber or nitrile gloves to protect skin from hypochlorite
- Use safety glasses or goggles in professional settings

Storage Best Practices
Keep bleach-based products and acid-based or ammonia-based products in separate, clearly labeled storage locations. In institutional or workplace settings, restrict access to incompatible chemicals and implement a sign-out or supervisory protocol for cleaning product use.
Proper storage separation works. CDC case reports from state hospitals found that institutional controls and storage separation prevented repeat chlorine gas exposures after initial incidents.
Personal Protective Equipment (PPE)
PPE requirements vary by setting. At minimum, everyone handling bleach-based products should use:
- Gloves: Rubber or nitrile for household use; chemical-resistant gloves for industrial or institutional settings
- Eye protection: Safety glasses for general use; sealed goggles when splashing is likely
- Respiratory protection: Required in confined or poorly ventilated spaces — an N95 is a baseline; full respirators for enclosed professional environments
Open windows and avoid face-level exposure whenever you apply bleach-based products, even in home settings.
What to Do If You're Exposed to Bleach Fumes
If you experience any symptoms of toxic gas exposure, act immediately. There is no antidote for chlorine or chloramine poisoning — fast action determines outcome.
Immediate response steps:
- Leave the contaminated area at once and move to fresh air
- Open all windows and doors before anyone re-enters
- Flush eyes with water for at least 15–20 minutes if exposed
- Shower and change clothes if gas contacted skin or clothing
Decontamination and Monitoring
Inhaling steamy shower air can help soothe the airway after mild exposures. Call Poison Control at 1-800-222-1222 even for mild symptoms — they'll advise whether you need medical evaluation.
When to Seek Emergency Care
Go to an emergency room or call 911 if symptoms include:
- Severe difficulty breathing
- Persistent chest pain
- Vomiting
- Loss of consciousness
- Symptoms that worsen rather than improve after reaching fresh air
Do not re-enter the space without confirming it has been fully ventilated and cleared.
Common Mistakes That Lead to Bleach Mixing Accidents
Most bleach-related accidents don't happen in labs or industrial settings — they happen at home, during routine cleaning. Understanding where people go wrong is the first step to avoiding a dangerous reaction.
Assuming "Natural" or "Gentle" Cleaners Are Safe to Mix
Vinegar is an acid, and many eco-friendly products contain acidic or ammonia-based ingredients. "Natural" labeling does not indicate chemical compatibility with bleach. Always check the ingredient list, not just the product packaging.
Sequential Cleaning Without Rinsing
One of the most common causes of accidental chlorine gas exposure is cleaning a surface with an acid-based product (like toilet bowl cleaner) and then applying bleach to the same surface without flushing it first. The residue is enough to trigger a dangerous reaction.
Ignoring Early Warning Signs
Many people dismiss early exposure symptoms as minor irritants and keep cleaning — which significantly increases their dose. Stop immediately if you notice any of the following:
- A sharp, acrid, or unfamiliar chemical odor
- Sudden eye watering or burning
- An unexplained cough or throat irritation
Leave the area right away, get fresh air, and ventilate the space before re-entering. Extended exposure — even at low concentrations — can escalate quickly.
Frequently Asked Questions
How toxic is chloramine gas?
Chloramine gas causes respiratory irritation, chest tightness, and nausea. Significant exposure can lead to pneumonia or fluid in the lungs — it's less volatile than chlorine gas but still capable of causing serious injury.
What gas forms when urine mixes with chlorine?
Urine contains ammonia, so when bleach contacts urine, chloramine gas is produced. This is why cleaning toilets or litter boxes with bleach without rinsing first can create hazardous exposure in an enclosed bathroom.
What mixtures create chlorine gas?
Chlorine gas forms when bleach or any hypochlorite-containing product is mixed with an acid. Common culprits include vinegar, toilet bowl cleaners, drain cleaners, rust removers, and dishwasher detergents.
Are chlorine and chlorine gas the same?
Chlorine is a chemical element that exists as a gas at room temperature. Chlorine gas specifically refers to its diatomic gas form (Cl₂), which is the toxic substance released when bleach is mixed with acids. The dissolved form in water (hypochlorite) is what gives bleach its disinfecting properties.
How toxic is chlorine gas?
Chlorine gas causes irritation at concentrations as low as 1 ppm, severe respiratory distress at moderate levels, and can be fatal at high concentrations. Even brief exposure in a poorly ventilated space like a bathroom can warrant an emergency room visit.
What is chlorine gas made of?
Chlorine gas (Cl₂) consists of two chlorine atoms bonded together. When sodium hypochlorite (bleach) reacts with an acid, the hypochlorite ion breaks down and releases Cl₂ along with water.


